Tips & Tricks Transfection
FAQ Transfection
Free Sample Offer
Transfection Reagent Selection Guide
Products Transfection
In cell biology, transfection is the term given to the introduction of foreign genetic material /DNA or RNA) into eukaryotic cells. In addition, a distinction is made between temporary introduction into the host cell (transient transfection) and permanent integration into the genome (stable transfection). The process of transfection basically corresponds to that of bacterial transformation, albeit under a different name.
Transfection methods
A variety of methods are used to introduce genetic material into eukaryotic cells:
Viral transfection uses genetically modified viruses which are no longer pathogenic and carry the gene to be introduced. In this method problems include the frequently non-directional stable integration of the gene into the genome of the host cell, the laborintensive production of the viruses and the strong immunological response.
Physical methods such as electroporation or microinjection are cost-intensive and of limited use in routine procedures.
Chemical methods include calcium phosphate precipitation, transfection using cationic polymers and - among the most efficient methods – lipofection, using cationic lipids. The Transfection reagents of Biontex (DOTAP, METAFECTENE®, METAFECTENE® PRO, METAFECTENE® SI, K2® Transfection System, K4® Transfection System) belong to the last group.

Transfection with cationic lipids
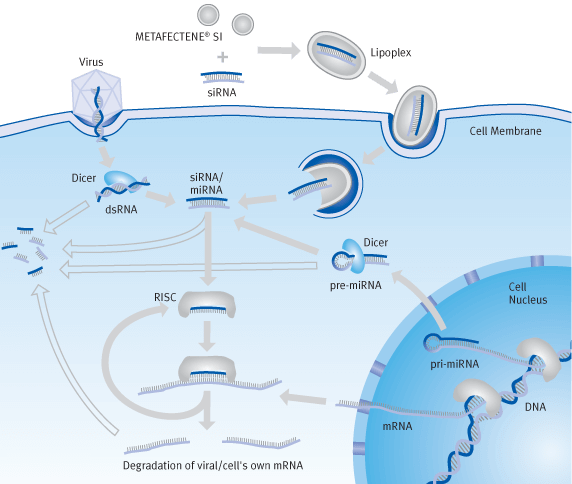
In aqueous solutions, cationic lipids form vesicles with a bilayer lipid sheet, known as liposomes. When liposomes encounter nucleic acids they re-form into nucleic acid lipid complexes called lipoplexes which can be actively taken up by eukaryotic cells by means of endocytosis. In this case, the lipoplex enters into the cell cytosol via the endosomes.

The endosomal structure is destroyed by increasing the osmotic pressure created by the lipids' buffering action within the endosomes and by the fusion of the lipid with the endosomal membrane. The ability of a lipid to destroy endosomes is one of the main characteristics of an efficient transfection reagent.
Transfection of DNA
DNA which is introduced into the cell cytosol cannot penetrate the membrane of the cell nucleus ("nuclear barrier"). Access to the nucleus is thus only possible if the nuclear membrane dissolves during mitosis. For this reason, the cell division rate is critical in DNA transfection and must be as high as possible for efficient transfection.
Transfection of RNA (siRNA/miRNA/mRNA)
For transfection of RNA their is no "nuclear barrier" as it is not neccessary for RNA to enter the nucleus for unfolding their biological effects. As a consequence the rate of cell division doesn't play a role.
Lipofection is a complex process in which the lipoplex amount and proportion of genetic material in relation to transfection reagent, i.e.lipoplex composition must be precisely optimized for each cell type.
Transfection reagents and methods show different profiles of performance in dependance of kind of nucleic acid and cell type used. Without reference, it is not possible to predict the suitability of a reagent or method concerning a special application and has to be determined empirically. Our "Transfection Reagent Selection Guide" supports you in the selection of a suitable transfection reagent for your application. We also offer samples for free, so you can test whether one of our transfection reagents is suitable for your purposes, without having to bear any costs.
Transfection mechanism
Even today, the complex process of lipofection is not fully understood. The following simplified outline shows the generally recognized stages of transfection:
- 1. Transfection-active lipoplexes spontaneously form from cationic lipids and negatively charged DNA
- 2. Lipoplexes are taken up by endocytosis
- 3. The DNA is released by destruction (osmotic effects and fusion) of the endosome membrane
- 4. The DNA enters the cell nucleus during mitosis (not in RNA transfection)

RNA Interference
RNA Interference (RNAi) is a natural regulatory mechanism in gene expression, controlled by short-chain RNA with approx. 21 – 28 nucleotides which are complementary to the mRNA of a target protein and can be found in all eukaryotic cells.
A distinction is made here between siRNA and miRNA, which is formed in the case of siRNA from doublestrand RNA (e.g. endogenic from transposons or exogenic from viruses) and, in the case of miRNA, from hairpin-structure endogenic RNA (from own pri-miRNA genes) by means of enzymatic cleavage with an endoribonuclease (Dicer).
These small RNA strands are bound in the cytosol by a ribonucleoprotein complex – the RISC complex (RNA induced silencing complex), the RNA double strand is separated and the leading strand is presented. When a complementary mRNA strand binds to the leading strand, it is broken down or deactivated by the RISC complex, thus preventing expression of the target protein and down-regulating the corresponding gene (knockdown).
The discovery of this mechanism, known as "gene silencing", has been a major research milestone in recent years, and is a powerful tool in the field of proteomics and research into functional relationships between genes.